-
1 phase electrodes
1) Электроника: электроды одной фазы, электроды фазы2) Макаров: электроды одной фазы (в ПЗС), электроды фазы (в ПЗС) -
2 phase electrodes
-
3 phase electrodes
The New English-Russian Dictionary of Radio-electronics > phase electrodes
-
4 phase electrodes
электроды одной фазы; электроды фазыEnglish-Russian dictionary of electronics > phase electrodes
-
5 electrode
- active electrode
- after-acceleration electrode
- applicator electrode
- base electrode - bipolar electrode
- body electrode
- calomel electrode
- calomel reference electrode
- carry electrode
- central electrode
- chronically implanted electrode
- collecting electrode
- collector electrode
- consumable electrode
- control electrode
- convergence electrode
- coplanar electrodes
- decelerating electrode
- decinormal calomel electrode
- deflection electrode
- double-skeleton electrode
- drain electrode
- edge electrode
- elevated electrode
- emitter electrode
- evaporated electrode
- exploring electrode
- face electrode
- floating electrode
- focusing electrode
- fuel electrode
- gas-diffusion electrode
- gas-free electrode
- gas-loaded electrode
- gate electrode
- ground electrode
- grounding electrode
- guard-ring electrode
- guide electrode
- horizontal-deflection electrodes
- hydrogen electrode
- ignitor electrode
- input electrode
- intensifier electrode
- intracellular electrode
- ion-selective electrode
- keep-alive electrode
- main electrode
- make-break electrode
- micro glass electrode
- modulating electrode
- monitoring electrode
- monopolar electrode
- multiple electrode
- negative electrode
- normal electrode
- output electrode
- oxidant electrode
- oxygen electrode
- pad electrode
- phase electrodes
- pickup electrode
- poling electrode
- porous electrode
- positive electrode
- post-accelerating electrode
- post-deflection accelerating electrode
- primer electrode
- probe electrode
- quinhydrone electrode
- radial deflecting electrode
- ray-control electrode
- RC electrode
- receiver electrode
- reference electrode
- reflecting electrode
- reflector electrode
- repelling electrode
- saturated calomel electrode
- segmented electrode
- self-aligned electrodes
- sensing electrode
- signal electrode
- skewed electrodes
- sounding electrode
- source electrode
- staggered electrodes
- standard electrode
- starter electrode
- starting electrode
- storage electrode
- superconducting electrode
- suspension electrode
- target electrode
- transfer electrode
- transparent electrode
- trigger electrode
- valve electrode
- vertical-deflection electrodes -
6 electrode
- active electrode
- after-acceleration electrode
- applicator electrode
- base electrode
- beam-forming electrode
- bifunctional electrode
- bipolar electrode
- body electrode
- calomel electrode
- calomel reference electrode
- carry electrode
- central electrode
- chronically implanted electrode
- collecting electrode
- collector electrode
- consumable electrode
- control electrode
- convergence electrode
- coplanar electrodes
- decelerating electrode
- decinormal calomel electrode
- deflection electrode
- double-skeleton electrode
- drain electrode
- edge electrode
- elevated electrode
- emitter electrode
- evaporated electrode
- exploring electrode
- face electrode
- floating electrode
- focusing electrode
- fuel electrode
- gas-diffusion electrode
- gas-free electrode
- gas-loaded electrode
- gate electrode
- ground electrode
- grounding electrode
- guard-ring electrode
- guide electrode
- horizontal-deflection electrodes
- hydrogen electrode
- ignitor electrode
- input electrode
- intensifier electrode
- intracellular electrode
- ion-selective electrode
- keep-alive electrode
- main electrode
- make-break electrode
- micro glass electrode
- modulating electrode
- monitoring electrode
- monopolar electrode
- multiple electrode
- negative electrode
- normal electrode
- output electrode
- oxidant electrode
- oxygen electrode
- pad electrode
- phase electrodes
- pickup electrode
- poling electrode
- porous electrode
- positive electrode
- post-accelerating electrode
- post-deflection accelerating electrode
- primer electrode
- probe electrode
- quinhydrone electrode
- radial deflecting electrode
- ray-control electrode
- RC electrode
- receiver electrode
- reference electrode
- reflecting electrode
- reflector electrode
- repelling electrode
- saturated calomel electrode
- segmented electrode
- self-aligned electrodes
- sensing electrode
- signal electrode
- skewed electrodes
- sounding electrode
- source electrode
- staggered electrodes
- standard electrode
- starter electrode
- starting electrode
- storage electrode
- superconducting electrode
- suspension electrode
- target electrode
- transfer electrode
- transparent electrode
- trigger electrode
- valve electrode
- vertical-deflection electrodesThe New English-Russian Dictionary of Radio-electronics > electrode
-
7 Héroult, Paul Louis Toussaint
SUBJECT AREA: Metallurgy[br]b. 1863 Thury-Harcourt, Caen, Franced. 9 May 1914 Antibes, France[br]French metallurigst, inventor of the process of aluminium reduction by electrolysis.[br]Paul Héroult, the son of a tanner, at the age of 16, while still at school in Caen, read Deville's book on aluminium and became obsessed with the idea of developing a cheap way of producing this metal. After his family moved to Gentillysur-Bièvre he studied at the Ecole Sainte-Barbe in Paris and then returned to Caen to work in the laboratory of his father's tannery. His first patent, filed in February and granted on 23 April 1886, described an invention almost identical to that of C.M. Hall: "the electrolysis of alumina dissolved in molten cryolite into which the current is introduced through suitable electrodes. The cryolite is not consumed." Early in 1887 Héroult attempted to obtain the support of Alfred Rangod Pechiney, the proprietor of the works at Salindres where Deville's process for making sodium-reduced aluminium was still being operated. Pechiney persuaded Héroult to modify his electrolytic process by using a cathode of molten copper, thus making it possible produce aluminium bronze rather than pure aluminium. Héroult then approached the Swiss firm J.G.Nehe Söhne, ironmasters, whose works at the Falls of Schaffhausen obtained power from the Rhine. They were looking for a new metallurgical process requiring large quantities of cheap hydroelectric power and Héroult's process seemed suitable. In 1887 they established the Société Metallurgique Suisse to test Héroult's process. Héroult became Technical Director and went to the USA to defend his patents against those of Hall. During his absence the Schaffhausen trials were successfully completed, and on 18 November 1888 the Société Metallurgique combined with the German AEG group, Oerlikon and Escher Wyss, to establish the Aluminium Industrie Aktiengesellschaft Neuhausen. In the early electrolytic baths it was occasionally found that arcs between the bath surface and electrode could develop if the electrodes were inadvertently raised. From this observation, Héroult and M.Killiani developed the electric arc furnace. In this, arcs were intentionally formed between the surface of the charge and several electrodes, each connected to a different pole of the AC supply. This furnace, the prototype of the modern electric steel furnace, was first used for the direct reduction of iron ore at La Praz in 1903. This work was undertaken for the Canadian Government, for whom Héroult subsequently designed a 5,000-amp single-phase furnace which was installed and tested at Sault-Sainte-Marie in Ontario and successfully used for smelting magnetite ore.[br]Further ReadingAluminium Industrie Aktiengesellschaft Neuhausen, 1938, The History of the Aluminium-Industrie-Aktien-Gesellschaft Neuhausen 1888–1938, 2 vols, Neuhausen.C.J.Gignoux, Histoire d'une entreprise française. "The Hall-Héroult affair", 1961, Metal Bulletin (14 April):1–4.ASDBiographical history of technology > Héroult, Paul Louis Toussaint
-
8 electrode
електрод - CCD electrode
- collector electrode
- control electrode
- coplanar electrodes
- depletion-mode gate electrode
- drain electrode
- enhancement-mode-gate electrode
- extra electrode
- gate electrode
- insulated gate electrode
- metal gate electrode
- phase electrode
- planar electrode
- reference electrode
- Schottky-barrier gateelectrode
- Schottkyelectrode
- self-aligned electrode
- source electrode
- spaced electrodes
- storage electrodeEnglish-Ukrainian dictionary of microelectronics > electrode
-
9 distributed neutral
система с нулевым рабочим проводником
-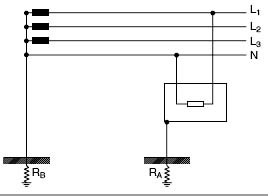
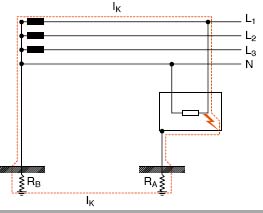
Рис. ABB
Система ТТПараллельные тексты EN-RU In TT systems the neutral and the exposed-conductiveparts are connected to earth electrodes electrically independent; therefore the earth fault current returns to the power supply node through the soil.
In this type of electrical installations the neutral is usually distributed and its function is making the phase voltage (e.g. 230 V) available for the supply of the single-phase loads of civil installations.
[ABB]В системах TT нулевой рабочий проводник и открытые проводящие части присоединены к разным заземляющим электродам, поэтому ток замыкания на землю возвращается к источнику питания через землю.
В электроустановках данного типа для распределения электроэнергии обычно применяют нулевой рабочий проводник, который совместно с линейными проводниками используется для питания однофазных нагрузок электроустановок жилых и общественных зданий напряжением, например, 230 В.
[Перевод Интент]For 3-pole circuit breakers used on 3-wire systems ( neutral not distributed), always set this value to 33 (see below) to avoid indications of a meaningless phase-to-neutral voltage.
[Schneider Electric]Если 3-полюсный автоматический выключатель используется в 3-проводной сети (т. е. без нулевого рабочего проводника), обязательно установите для этого параметра значение 33 (порядок настройки описан ниже), это исключит отображение параметра «фазное напряжение», не имеющего смысла в данном случае.
[Перевод Интент]Тематики
EN
Англо-русский словарь нормативно-технической терминологии > distributed neutral
-
10 Bacon, Francis Thomas
SUBJECT AREA: Aerospace[br]b. 21 December 1904 Billericay, Englandd. 24 May 1992 Little Shelford, Cambridge, England[br]English mechanical engineer, a pioneer in the modern phase of fuel-cell development.[br]After receiving his education at Eton and Trinity College, Cambridge, Bacon served with C.A. Parsons at Newcastle upon Tyne from 1925 to 1940. From 1946 to 1956 he carried out research on Hydrox fuel cells at Cambridge University and was a consultant on fuel-cell design to a number of organizations throughout the rest of his life.Sir William Grove was the first to observe that when oxygen and hydrogen were supplied to platinum electrodes immersed in sulphuric acid a current was produced in an external circuit, but he did not envisage this as a practical source of electrical energy. In the 1930s Bacon started work to develop a hydrogen-oxygen fuel cell that operated at moderate temperatures and pressures using an alkaline electrolyte. In 1940 he was appointed to a post at King's College, London, and there, with the support of the Admiralty, he started full-time experimental work on fuel cells. His brief was to produce a power source for the propulsion of submarines. The following year he was posted as a temporary experimental officer to the Anti-Submarine Experimental Establishment at Fairlie, Ayrshire, and he remained there until the end of the Second World War.In 1946 he joined the Department of Chemical Engineering at Cambridge, receiving a small amount of money from the Electrical Research Association. Backing came six years later from the National Research and Development Corporation (NRDC), the development of the fuel cell being transferred to Marshalls of Cambridge, where Bacon was appointed Consultant.By 1959, after almost twenty years of individual effort, he was able to demonstrate a 6 kW (8 hp) power unit capable of driving a small truck. Bacon appreciated that when substantial power was required over long periods the hydrogen-oxygen fuel cell associated with high-pressure gas storage would be more compact than conventional secondary batteries.The development of the fuel-cell system pioneered by Bacon was stimulated by a particular need for a compact, lightweight source of power in the United States space programme. Electro-chemical generators using hydrogen-oxygen cells were chosen to provide the main supplies on the Apollo spacecraft for landing on the surface of the moon in 1969. An added advantage of the cells was that they simultaneously provided water. NRDC was largely responsible for the forma-tion of Energy Conversion Ltd, a company that was set up to exploit Bacon's patents and to manufacture fuel cells, and which was supported by British Ropes Ltd, British Petroleum and Guest, Keen \& Nettlefold Ltd at Basingstoke. Bacon was their full-time consultant. In 1971 Energy Conversion's operation was moved to the UK Atomic Energy Research Establishment at Harwell, as Fuel Cells Ltd. Bacon remained with them until he retired in 1973.[br]Principal Honours and DistinctionsOBE 1967. FRS 1972. Royal Society S.G. Brown Medal 1965. Royal Aeronautical Society British Silver Medal 1969.Bibliography27 February 1952, British patent no. 667,298 (hydrogen-oxygen fuel cell). 1963, contribution in W.Mitchell (ed.), Fuel Cells, New York, pp. 130–92.1965, contribution in B.S.Baker (ed.), Hydrocarbon Fuel Cell Technology, New York, pp. 1–7.Further ReadingObituary, 1992, Daily Telegraph (8 June).A.McDougal, 1976, Fuel Cells, London (makes an acknowledgement of Bacon's contribution to the design and application of fuel cells).D.P.Gregory, 1972, Fuel Cells, London (a concise introduction to fuel-cell technology).GW
См. также в других словарях:
Dépôt chimique en phase vapeur assisté par plasma — Equipement de PECVD. Le dépôt chimique en phase vapeur assisté par plasma (ou PECVD, pour Plasma Enhanced Chemical Vapor Deposition en anglais) est un procédé utilisé pour déposer des couches minces sur un substrat à partir d un état gazeux… … Wikipédia en Français
Chromatographe en phase gazeuse — Chromatographie en phase gazeuse La chromatographie en phase gazeuse (CPG) est, comme toutes les techniques de chromatographie, une technique qui permet de séparer des molécules d un mélange éventuellement très complexe de nature très diverses.… … Wikipédia en Français
Chromatographie En Phase Gazeuse — La chromatographie en phase gazeuse (CPG) est, comme toutes les techniques de chromatographie, une technique qui permet de séparer des molécules d un mélange éventuellement très complexe de nature très diverses. Elle s applique principalement aux … Wikipédia en Français
Chromatographie en Phase Gazeuse — La chromatographie en phase gazeuse (CPG) est, comme toutes les techniques de chromatographie, une technique qui permet de séparer des molécules d un mélange éventuellement très complexe de nature très diverses. Elle s applique principalement aux … Wikipédia en Français
Chromatographie en phase gazeuse — La chromatographie en phase gazeuse (CPG) est, comme toutes les techniques de chromatographie, une technique qui permet de séparer des molécules d un mélange éventuellement très complexe de nature très diverses. Elle s applique principalement aux … Wikipédia en Français
Josephson phase — In superconductivity, the Josephson phase is the difference of the phases of the quantum mechanical wave function in two superconducting electrodes forming a Josephson junction.For example, if the macroscopic wave functions Psi 1 and Psi 2 in… … Wikipedia
analysis — /euh nal euh sis/, n., pl. analyses / seez /. 1. the separating of any material or abstract entity into its constituent elements (opposed to synthesis). 2. this process as a method of studying the nature of something or of determining its… … Universalium
Crystal oscillator — A miniature 4 MHz quartz crystal enclosed in a hermetically sealed HC 49/US package, used as the resonator in a crystal oscillator. A crystal oscillator is an electronic oscillator circuit that uses the mechanical resonance of a vibrating crystal … Wikipedia
ÉLECTROPHYSIOLOGIE — Les êtres vivants, végétaux et animaux, sont communément le siège de phénomènes électriques intimement liés aux activités vitales, dont ils sont un des aspects les plus révélateurs. On met ces phénomènes en évidence à l’aide d’électrodes… … Encyclopédie Universelle
Spectre de masse — Spectrométrie de masse Pour les articles homonymes, voir MS. La spectrométrie de masse (mass spectrometry ou MS) est une technique physique d analyse permettant de détecter et d identifier des molécules d’intérêt par mesure de leur masse, et de… … Wikipédia en Français
Spectrometrie de masse — Spectrométrie de masse Pour les articles homonymes, voir MS. La spectrométrie de masse (mass spectrometry ou MS) est une technique physique d analyse permettant de détecter et d identifier des molécules d’intérêt par mesure de leur masse, et de… … Wikipédia en Français
